The internal resistance provides valuable information about a battery as high reading hints at end-of-life. This is especially true with nickel-based systems. Resistance measurement is not the only performance indicator as the value between batches of lead acid batteries can vary by 5–10 percent, especially with stationary units. Because of this wide tolerance, the resistance method works best when comparing the readings of a given battery from birth to retirement. Service crews are asked to take a snapshot of each cell or monoblock at time of installation and then measure the subtle changes as the cells age.
There is a notion that internal resistance is related to capacity, but this is false. The resistance of modern lead acid and lithium-ion batteries stays flat through most of the service life. Better electrolyte additives have reduced internal corrosion issues that affect the resistance. This corrosion is also known as parasitic reactions on the electrolyte and electrodes. Figure 1 shows capacity fade with cycling in relation to the internal resistance of Li-ion cells.
.jpg)
Resistance does not reveal the state-of-health of a battery and often stays flat with use and aging.
Cycle test on Li-ion batteries at 1C:
- Charge: 1,500mA to 4.2V, 25°C
- Discharge: 1,500 to 2.75V, 25°C
What Is Impedance?
Before exploring the different methods of measuring the internal resistance of a battery, let’s examine what electrical resistance means and understand the difference between pure resistance (R) and impedance (Z). R is pure resistance and Z includes reactive elements such as coils and capacitors. Both readings are obtained in ohms (Ω), a measurement that goes back to the German physicist Georg Simon Ohm, who lived from 1798 to 1854. (One ohm produces a voltage drop of 1V with a current of 1A.) The electric conductivity is also measured in siemens (s) that is reciprocal to ohmic values.
The electrical resistance of a pure load, such as a heating element, has no reactance. Voltage and current flow in unison and there is no advancing or trailing phase. The ohmic resistance is the same with direct current (DC) and alternating current (AC). The power factor (pf) is 1, providing the most accurate metering of the power consumed.
Most electrical loads are reactive and consist of capacitive reactance (capacitor) and inductive reactance (coil). The capacitive reactance decreases with higher frequency while the inductive reactance increases. An analogy of inductive reactance is an oil damper that stiffens when applying a fast back-and-forth action.
A battery has resistance, capacitance and inductance, and the term impedance includes all three in one model. Impedance can best be illustrated with the Randles model (Figure 2) that comprises resistors R1 and R2 as well as capacitor C. The inductive reactance is commonly omitted because it plays a negligible role in a battery, especially at a low frequency.
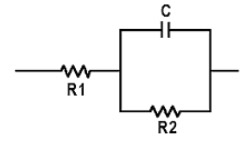
The overall battery resistance consists of ohmic resistance, as well as inductive and capacitive reactance. The diagram and electrical values differ for every battery.
- R1 = Internal resistant; R2 = Charge transfer
- C1 = Double layer capacitor
Measuring the battery by resistance is almost as old as the battery itself and several methods have developed over time, all of which are still in use.
DC Load Method
The ohmic measurement is one of the oldest and most reliable test methods. The battery receives a brief discharge for a second or longer. The load current for a small battery is 1A or less; for a starter battery it might be 50A or more. A voltmeter measures the open circuit voltage (OCV) with no load, followed by the second reading with a load; Ohm’s law calculates the resistance value (voltage difference divided by current equals resistance).
DC load measurements work well to check large stationary batteries, and the ohmic readings of the device are very accurate and repeatable. High-end test instruments claim resistance readings in the 10 micro-ohm range. Many garages use the carbon pile to measure starter batteries and an experienced mechanic gets a reasonably good assessment of the battery.
The DC load method has limitations in that it blends R1 and R2 of the Randles model into one combined resistor and ignores the capacitor (see Figure 3). “C” is an important component of a battery that represents 1.5 farads per 100Ah capacity. In essence, the DC method sees the battery as a resistor and can only provide ohmic references. In addition, the DC load method gets similar readings from a good battery that is partially charged and a marginal battery that is fully charged. State-of-charge and capacity estimations are not possible.
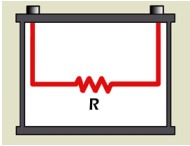
The true integrity of the Randles model cannot be seen. R1 and R2 appear as one ohmic value.
The two-tier DC load method offers an alternative method by applying two sequential discharge loads of different currents and time durations. The battery first discharges at a low current for 10 seconds, followed by a higher current for 3 seconds (see Figure 4); the Ohm’s law calculates the resistance values. Evaluating the voltage signature under the two load conditions offers additional information about the battery, but the values are strictly resistive and do not reveal SoC or capacity estimations. The load test is the preferred method for batteries that power DC loads.
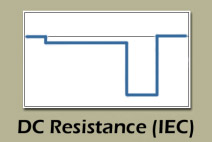
The two-tier DC load follows the IEC 61951-1:2005 standards and provides lifelike test conditions for many DC battery applications.
AC Conductance
Conductance measurement to evaluate starter batteries was first reported by Keith Champlin in 1975 by demonstrating a linear correlation between load test and conductance. When injecting a frequency of about 90 hertz, capacitive and inductive reactance converge with a 70–90Ah lead acid battery, resulting in a negligible voltage lag that minimizes the reactance. (This frequency rises with a smaller battery and drops with a large pack.) AC conductance meters are commonly used in car garages to measure CCA. The single-frequency method (Figure 5) sees the components of the Randles model as one complex impedance called the modulus of Z.
The individual components of the Randles model are molten together and cannot be distinguished.
The 1,000-hertz (Hz) ohm test is another common method. A 1,000Hz signal excites the battery and Ohm’s law calculates the resistance. Note that the AC method shows different values to the DC method when measuring a reactive resistance, and both readings are correct.
For example, Li-ion in an 18650 cell produces about 36mOhm with a 1,000Hz AC signal and roughly 110mOhm with a DC load. Since both readings are valid, yet far apart, the user must consider the application. The pulse DC load method provides valuable readings for a DC application such as a heating element or an incandescent light, while the 1,000Hz method better reflects the performance requirements of a digital load, such as portable computing and mobile phones that rely to a large extent on the capacitive characteristics of a battery. Figure illustrates the 1,000Hz-method. (See also Table 2 in BU-802a: How does Rising Internal Resistance affect Performance?)
The 1000-hertz provides reactive resistance readings. This has been the preferred method for taking impedance snapshots of batteries powering digital devices.
Electrochemical Impedance Spectroscopy (EIS)
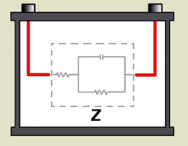
Research laboratories have been using EIS for many years to evaluate battery characteristics. High equipment cost, slow test times and the need for trained professionals to decipher the large volume of data have limited this technology to laboratory environments. EIS reads R1, R2 and C values in the Randles model (Figure 7); however, correlating the data into CCA and capacity estimations requires complex modeling(See BU-904: How to Measure Capacity)
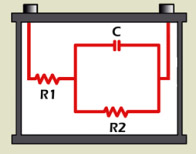
R1, R2 and C are measured separately, which enables state-of-charge and capacity measurements.
References
[1] Courtesy of Cadex

Comments
Looking for comments from the previous website?
Comments from the previous website are not compatible with our new commenting system but we have preserved them so our users can still reference and make use the information in them.
